
The outer energy level or “shell” is considered full when it has the maximum number of electrons that can fit into that level.Ģ6 How is the dispertion of electrons in shell? For example X Nitrogen N 7 X X N X X 14 X XĢ4 DOT & CROSS DIAGRAMS O Cl X X X X X X X X X Cl X X X X X X X O X X X XĨ 17 X O Cl a) b) X 35 X 16 X X X X X Cl X X X X X X X O X X X X X X X X X XĢ5 How is the dispertion of electrons in shell? With Dot & Cross diagrams elements and compounds are represented by Dots or Crosses to show electrons, and circles to show the shells. With electronic configuration elements are represented numerically by the number of electrons in their shells and number of shells For example Nitrogen configuration = 2, 5 7 2 in 1st shell 5 in 2nd shell N = 7 14 Mass number = 12 Atomic number = 6 p+ = 6 no = 6 e- = 6 6 p and 6 n live in the nucleus first shell a maximum of 2 electrons second shell a maximum of 8 electrons third shell a maximum of 8 electronsġ9 How exactly are the particles arranged?īohr Model of the atom: All of the protons and the neutrons The 3rd ring can hold up to 18 e- The 1st ring can hold up to 2 e- The 4th ring and any after can hold up to 32 e- The 2nd ring can hold up to 8 e. The energy levels of electrons are labeled by principal quantum numbers (n)ġ8 ATOMIC STRUCTURE Electrons are arranged in Energy Levels or Shells around the nucleus of an atom. (like rungs on a ladder) The amount of energy required to move an electron to another energy level is called a quantum (shell). The electrons have fixed energies called energy levels. The electron cloud is a cloud of varying density surrounding the nucleus.Įlectrons are found in specific circular paths (orbits) around the nucleus. Model of the atom pictures the electrons moving around the nucleus in a region called an electron cloud. The electrons are equal to the number of protons Ex: O has a mass of 16 and an atomic number of 8 and has 8 electrons. Since it has 8 proton it must have 8 neutrons Mass number: the number of protons and neutrons in the nucleus Ex: oxygene can have a mass of 16. 2 protons = He, 29 protons = Cuġ2 How do we know the number of subatomic particles in an atom? In an atom: The protons = the electrons If 20 protons are present in an atom then 20 electrons are there to balance the overall charge of the atom-atoms are neutral The neutrons have no charge ġ1 How do we know the number of subatomic particles in an atom?Ītomic number: this number indicates the number of protons in an atom Ex: Hydrogen’s atomic number is 1 So hydrogen has 1 proton Ex: Carbon’s atomic number is 6 So carbon has 6 protons **The number of protons identifies the atom. Protons and neutrons live compacted in the tiny positively charged nucleus They account most of the mass of the atom The negatively charged electrons are small and have a relatively small mass but occupy a large volume of space outside the nucleusġ0 How do the subatomic particles balance each other? The 3rd subatomic particle resides outside of the nucleus in the electron cloud Electron: the subatomic particle with a negative charge and relatively no mass 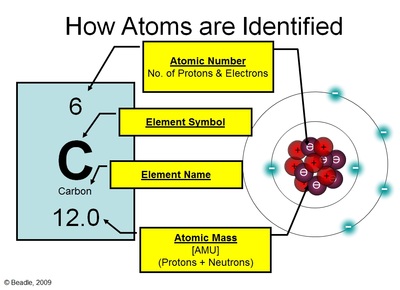
Nucleus: the center of the atom that contains the mass of the atom Electron cloud: region that surrounds the nucleus that contains most of the space in the atom Nucleus Electron Cloudĥ What’s in the Nucleus? The nucleus contains 2 of the 3 subatomic particles: Protons: positively charged subatomic particles Neutrons: neutrally charged subatomic particlesĦ HELIUM ATOM + - + - Shell proton neutron electronħ ATOMIC STRUCTURE Particle Charge Mass proton + charge 1 neutron No charge 1 electron - charge nil 2 Bohr’s Atom electrons in orbits nucleusģ What is an atom? Atom: the smallest unit of matter that retains the identity of the substanceĤ Atomic Structure Atoms are composed of 2 regions:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
